- Blog
- Playstation 2 bios
- Download lego ninjago tournament apk
- Looks like something went wrong while logging in remembear
- Cloudapp lifetime deal
- Basilisk ii mac os utilities
- Cyberpower powerpanel personal edition
- Connecting docear with zotero
- Ny times party quiz
- Hecta number
- Expedat droplets
- Stick rpg full screen
- Complement angle and supplementary angle
- Mus2 crack
- Thor movies in order
- #Connecting docear with zotero pdf
- #Connecting docear with zotero install
- #Connecting docear with zotero software
- #Connecting docear with zotero trial

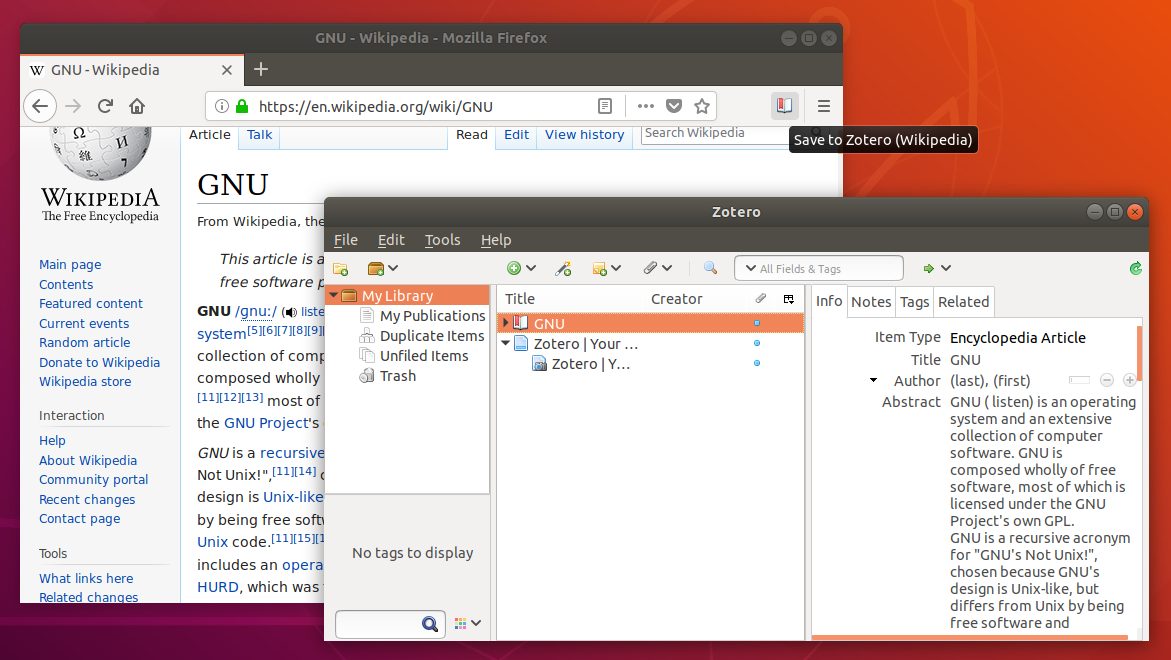
Even though Zotero does not have an on-demand customer support service, its Web site offers a wealth of information for users.
#Connecting docear with zotero install
Users can install a plug-in that allows Zotero to integrate with Microsoft Word. Sixteen citation styles are available when Zotero is initially downloaded, with many more freely available. Zotero offers more options for note taking than the better-known citation management system EndNote. View all references Zotero automatically imports citation information from a number of sources, including nonsubscription, newspaper, and commercial Web sites, and Web-based databases such as PubMed and MedlinePlus. Zotero: The Next-Generation Research Tool. 1 1.Ĭenter for History New Media, George Mason University. Zotero is a free and open source Firefox extension that exists within the Web browser and allows one to collect, manage, store, and cite resources in a single location. Ultimately, the choice of an RM should reflect the user's needs and work habits.
#Connecting docear with zotero pdf
The other RMs offered contrasting strengths: CiteULike in simplicity and social networking, Zotero in ease of automated importing, and Mendeley in PDF management. The authors found that RefWorks generated the most accurate citations.

To test citation accuracy, bibliographies of these references were generated in five different styles. To test importing and data management features, fourteen references from seven bibliographic databases were imported into each RM, using automated features whenever possible. The present study compares four prominent RMs: CiteULike, RefWorks, Mendeley, and Zotero, in terms of features offered and the accuracy of the bibliographies that they generate. Librarians are often called upon to provide support for these products.
#Connecting docear with zotero software
Reference management (RM) software is widely used by researchers in the health and natural sciences. and shall report the evaluation within three years. We are currently evaluating our protocol. The market phase describes best practices for medical device development for consumer usage. The product phase describes best practices for laboratory examination for medical device development in the research stage. The technology phase includes primary literature and lab review for technological units for the non-invasive medical device. This article describes in detail for each point for this protocol, using our current non-invasive blood glucose and Haemoglobin level medical device as an example. We have developed a three-phase protocol, consist of technology, product, and market phase.
This article is a preposition article developed from literature research and currently evaluated in three years of research in the consortium.
This protocol is currently evaluated by "Konsorsium Riset Alat Ukur Haemoglobin, Kadar Gula (Glukosa dan HbA1c) Non- Invasive" between IPB University, Agency for the Assessment and Application of Technology, and PT Tesena Inovindo.
#Connecting docear with zotero trial
We have combined best practices from Simplified Pressman Standard and Indonesian Technology Readiness Level and Clinical Trial Regulation as a base for our medical device development and evaluation methods. This article describes our current protocol for medical device development and evaluation. The new Indonesian national regulation for medical device evaluation necessitates the modification of current best practices for solution development.
- Blog
- Playstation 2 bios
- Download lego ninjago tournament apk
- Looks like something went wrong while logging in remembear
- Cloudapp lifetime deal
- Basilisk ii mac os utilities
- Cyberpower powerpanel personal edition
- Connecting docear with zotero
- Ny times party quiz
- Hecta number
- Expedat droplets
- Stick rpg full screen
- Complement angle and supplementary angle
- Mus2 crack
- Thor movies in order
